Chemistry Investigation——Enthalpy Change of a Displacement Reaction
Data Collection
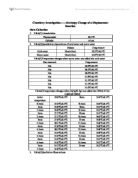
- Table(1):Uncertainties
- Table(2)Quantitative observations of cold water and warm water
Table(3)Temperature changes when warm water was added into cold water
Table(4)Temperature changes when Zn(5g±0.1g) was added into 100mL±1mL CuSO4(0.500M)
- Table(5)Qualitative Observations
Data processing and presentation
This experiment includes two parts, the determination of a calorimeter constant and then the enthalpy change of the reaction between Zn and CuSO4.
Part A
- The heat absorbed by the cold water.
a) The mass of cold water.
m(water)=ρ(water)×V(water)
=1g/mL×50mL
=50g
Absolute uncertainty=±1mL
(Assume that the absolute uncertainty of ρ(water) is negligible)
% uncertainty=1/50 × 100%=2%
b) The change of the temperature
△T is the temperature change after the warm water was added into the cold water.
△T=38.2℃-20.2℃
=18℃
Absolute uncertainty= ±0.5℃ + ±0.5℃=±1℃
% uncertainty=1℃/18℃ × 100%=6%
c) The heat absorbed by the cold water
Q(cold water)=m×s×△T
=50g×4.18J/g℃×18℃
=3.8×103J
=3.8kJ
% uncertainty=2% + 6%=8%
(Assume that the absolute uncertainty of the specific heat of water is negligible)

Absolute uncertainty=3.8kJ × 8%=±0.3kJ
- The heat released by the warm water.
a) The mass of warm water.
m(water)=ρ(water)×V(water)
=1g/mL×50mL
=50g
Absolute uncertainty=±1mL
(Assume that the absolute uncertainty of ρ(water) is negligible)
% uncertainty=1/50 × 100%=2%
b) The change of the temperature
△T is the temperature change after the warm water was added into the cold water.
△T=61.8℃-38.2℃
=23.6℃
Absolute uncertainty=±0.5℃ + ±0.5℃=±1℃
% uncertainty=1℃/23.6℃ × 100%=4%
c) The heat released by the warm water
Q(warm water)=m×s×△T
=50g×4.18J/g℃×23.6℃
=4.9×103J
=4.9kJ
% uncertainty=2% + 4%=6%
(Assume that the absolute uncertainty of the specific heat of water is negligible)
Absolute uncertainty=4.9 × ...
This is a preview of the whole essay
Absolute uncertainty=3.8kJ × 8%=±0.3kJ
- The heat released by the warm water.
a) The mass of warm water.
m(water)=ρ(water)×V(water)
=1g/mL×50mL
=50g
Absolute uncertainty=±1mL
(Assume that the absolute uncertainty of ρ(water) is negligible)
% uncertainty=1/50 × 100%=2%
b) The change of the temperature
△T is the temperature change after the warm water was added into the cold water.
△T=61.8℃-38.2℃
=23.6℃
Absolute uncertainty=±0.5℃ + ±0.5℃=±1℃
% uncertainty=1℃/23.6℃ × 100%=4%
c) The heat released by the warm water
Q(warm water)=m×s×△T
=50g×4.18J/g℃×23.6℃
=4.9×103J
=4.9kJ
% uncertainty=2% + 4%=6%
(Assume that the absolute uncertainty of the specific heat of water is negligible)
Absolute uncertainty=4.9 × 6%=±0.3kJ
- The heat absorbed by the calorimeter
According to the principle of the conservation of energy, the heat released by the warm water should be equal to the heat absorbed by the cold water and the calorimeter, hence:
Q warm water=Q cold water +Q calorimeter
Therefore, the equation can be converted into:
Qcalorimeter=Q warm water-Q cold water
=4.9kJ-3.8kJ
=1.1kJ
Absolute uncertainty=±0.3kJ + ±0.3kJ=±0.6 kJ
% uncertainty=0.6/1.1 × 100%=54%
- The calorimeter constant
Qcalorimeter can be written as:
Qcalorimeter=Ccalorimeter×△T
In which △T is the temperature change undergone by the calorimeter(the △T of cold water), and Ccalorimeter is the calorimeter constant, which represents the number of kilo-joules of heat required to warm the calorimeter by 1℃.
a) The change of the temperature
△T=38.2℃-20.2℃
=18℃
Absolute uncertainty= ±0.5℃ + ±0.5℃=±1℃
% uncertainty=1℃/18℃ × 100%=6%
b)The calorimeter constant
Ccalorimeter=Qcalorimeter/△T
=1.1kJ/18℃
=0.061kJ/℃
=61J/℃
Absolute uncertainty=54% + 6%=60%
% uncertainty=0.061kJ/℃×54%=0.033kJ
Part B
In this part, the enthalpy change of the reaction between Zn and CuSO4 would be calculated.
To do this, the graph of the temperature changes during the reaction is drawn below:
In order to calculate the enthalpy change of the reaction, the heat absorbed or released is needed. According to the graph, it can be seen that the reaction is exothermic due to rising in the temperature. Therefore, the heat released by the reaction needs to be calculated.
- The heat released by the reaction
a)The mass of water.
m(water)=ρ(water)×V(water)
=1g/ml × 100ml
=100g
Absolute uncertainty=±1mL
(Assume that the absolute uncertainty of ρ(water) is negligible)
% uncertainty=1/100 × 100%=1%
b)The change of the temperature
The intersection of the two lines in the graph should be the highest temperature that would have been reached if no heat is lost to surroundings. Approximately the temperature should be 38.5℃
△T=38.5℃-19.8℃
=18.7℃
Absolute uncertainty=±0.5℃ + ±0.5℃=±1℃
% uncertainty=1/18.7 × 100%=5%
c)The heat absorbed by the solution.
(Assume the Ssolution=Swater=4.18J/ g℃)
Heat released=m×s×△T
=100g×4.18 J/g℃×18.7℃
=7.81×103J
=7.8kJ
% uncertainty=1%+5%=6%
(Assume that the absolute uncertainty of the specific heat of water is negligible)
Absolute uncertainty=7.8kJ×6%=0.5kJ
- The heat energy absorbed by the calorimeter.
Qcalorimeter=Ccalorimeter×△T
Qcalorimeter=61J/℃×18.7℃=1.1×103J=1.1kJ
% uncertainty=60%+5%=65%
Absolute uncertainty=1.1kJ×65%=0.71kJ
- The total heat released by the reaction
In this reaction, only using the data obtained in this experiment to calculate the heat absorbed by the solution is not accurate. Because the calorimeter was not completely insulated, it absorbed some heat from the reaction. Therefore the total heat released by the reaction should also include the heat absorbed by the calorimeter.
The total heat energy=Qwater+Qcalorimeter
=7.81kJ +1.1kJ
=8.9kJ
Absolute uncertainty=±0.5kJ + ±0.71kJ=±1.2kJ
% uncertainty=1.2/8.9 × 100%=13%
- The enthalpy change of the reaction
The heat will vary with the amount of the limiting reagent, so by convention it is given for molar amounts in the equation as it is written.
Therefore, △H=the heat absorbed or released/the number of moles=Q/n
In the reaction between CuSO4 and Zn, the limiting reagent needs to be determined.
- The limiting reagent
The chemical formula of the reaction is:
Zn(s) + CuSO4(aq)==> Cu(s) + ZnSO4(aq)
The amount of CuSO4=0.500M×0.100L=0.0500mol
The amount of Zn=m/M=5.0g/65.39gmol-1=0.076mol
According to the balanced equation, the molar ratio of Zn and CuSO4 should be 1 : 1, but here the amount of Zn is more than that of CuSO4, thus Zn was excessive in the reaction and CuSO4 is the limiting reagent.
- The amount of the limiting reagent
Since the limiting reagent is CuSO4, the number of moles of it needs to be taken.
The amount of CuSO4=0.500M×0.100L=0.0500mol
Absolute uncertainty=±1mL
% uncertainty of the volume=1mL/100mL×100%=1%
% uncertainty of the concentration of CuSO4 is 0.51% (given)
The total percentage uncertainty =1%+0.51%=1.51%
- The enthalpy change of the reaction
△H=Q/n=8.9kJ/0.0500mol=178kJ/mol ≈ 1.8×102kJ/mol
% uncertainty=13%+1.51%=14.51%
Absolute uncertainty=1.8×102kJ/mol×14.51%=26kJ/mol=30kJ/mol
Since the reaction is exothermic, there should be a minus sign in front of the enthalpy change, hence △H=-1.8×102kJ/mol
Therefore the enthalpy change of the reaction between Zn and CuSO4 is
(1.8±0.3)×102kJ/mol
Conclusion and evaluation:
- In conclusion, the enthalpy change of the reaction between Zn and CuSO4 is
-178kJ/mol ± 28kJ/mol. The literature value of the enthalpy change is
-218kJ/mol. By comparison, the percentage error can be got.
From Data processing and presentation, it is known that the percentage uncertainty of this experiment is 14.51% which is less than the percentage error 18.3%. However, the result is acceptable since it is close to the literature value.
- Limitations
Since the % uncertainty is less than the error, random errors alone cannot account for the difference between the experimental value and the literature value. Systematic errors must have contributed to this difference.
- Systematic errors
- To compensate for the heat lost by the water in this reaction to the surroundings as the reaction proceeds a plot of temperature against time can be drawn. By extrapolating the graph, the temperature rise that would have been reached if no heat is lost can be figured out. But here, it is assumed that the rate at which the reaction was cooling off is the same. Hence the temperature the reaction should reach might be found accurately. The value I got can be higher or lower and this leads to the systematic error to happen
- From the graph, it can be seen that the line is not smooth, it sometimes rises and falls. This is because that the solution was not well mixed and stirred. The zinc powder might be lumping when it was put into the calorimeter. After the zinc powder was added, the calorimeter needed to be stirred in order to let it react with CuSO4. Hence different rate of stirring can affect the rate of reaction and temperature.
- Although the calorimeter does not conduct heat well, it does still absorb some heat. In addition, a small quantity of heat may be transferred to or from the copper wire used for stirring the calorimeter’s contents or to the glass of the thermometer used to measure temperature changes. Some heat may also be lost through the openings of the calorimeter. All of the above may let the calorimeter constant I got in Part A be much smaller since the reaction was very slow in Part B and much more heat was lost to the surroundings.
- Although systematic errors affect the experiment greatly, random error still have its influence. When I calculate the calorimeter constant, the uncertainty I got is even bigger than 50%. This is due to the inaccuracy of the thermometer which made △T too small. If △T is too small, the heat absorbed by the calorimeter would be small. But in contrast, the absolute uncertainty is comparatively bigger, this causes the total uncertainty to be very great.
- Sometimes the error happens in the procedure of calculating. In the calculation of the calorimeter constant in this experiment, the whole process can be combined into one big and complex formula which is:
Ccalorimeter=(ms△Twarm water-ms△Tcold water)/△Tcold water
=ms[(△Twarm water-△Tcold water)/ △Tcold water
However, in order to present the data more clearly, I have used several steps to calculate the calorimeter constant. This would accumulate the errors since I have calculated ms△T twice. For this reason, the error I got is bigger.
- Improvements
- During the reaction, there was still a lot of heat lost to the surroundings. To prevent this, next time, I can use plastic or glass stick as the stirring rod since they can conduct less heat than the copper wire. Moreover, next time, cotton needs to be used to fill the openings on the calorimeter. Towel is also necessary because it is needed to wrap the calorimeter to prevent the heat lost.
- The time required for the reaction needs to be extended. By convention, the temperature would gradually fall as there is more heat lost to the surroundings. But according to the graph, the line is still very smooth, this means that 17 minutes is not enough. If the time is extended, the graph can be plotted more accurately and the extrapolating line can point to a more accurate temperature value.
- Next time, I should use more combined formulae. Since the more steps you use, the bigger the errors you have. The number of the steps should be reduced.
- Using a more accurate thermometer since the accuracy of the thermometer is the main reason that causes the % uncertainty to be so high.