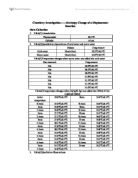
Enthalpy. This experiment's aim is to find out, enthalpy change of a reaction, and to determine whether it is exothermic or endothermic.
Lab report on Emthapy
Aim :
This experiment's aim is to find out, enthalpy change of a reaction, and to determine whether it is exothermic or endothermic.
Apparatus :
Measuring Trays
Weighing Scale
Citric acid
Sodium Carbonate
Polystyrene cup
Thermometer
Reaction beaker + lid
Copper Sulphate
Zinc (powdered)
Stopwatch
Method:
- Pour 25 ml of Copper Sulphate into the reaction beaker.
- Place the reaction beaker in the polystyrene cup and put the thermometer in the cap.
- Record the temperature of the solution.
- Add the 0.5 grams of Zinc powder and close the lid immediately.
- Record Maximum change of temperature.
- Repeat steps 1 – 5 but this time use citric acid in place of Copper Sulphate and 3 grams of Sodium Carbonate instead of zinc.
- Calculate energy change for the amounts of substance used.
- Calculate the amount of the reactant in solution in the cup.
- For each reaction calculate energy change per mole of the reactant which was in solution at the start.
- Write a symbol equation for the first reaction and a word equation for the second one.
- Write the energy change per mole of citric acid.
- Draw an energy level diagram for the enthalpy change of both reactions.