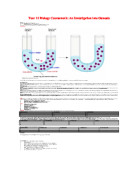
Osmosis is also present within animal cells however it is different. Animal cells do not have cell walls and so the amount of water going into a cell must be carefully controlled otherwise if the animal cell has too much water it will eventually bursts which means if it doesn’t have enough water than it will collapse.
The factors, which may affect osmosis, are temperature, concentration of a solution and pressure. I have chosen to investigate concentration, as this is the most practical to measure. I will obtain my results by the loss or gain in the weight of a potato chip which has been placed in a solution varying in concentration from 0 moles to 1 moles. I have decided to obtain my results by the gain or loss in weight, as it will not be a fair test if I measure the length. I measured the length in my preliminary experiment however this is inaccurate because when I cut a potato chip I might slice the top, therefore I could measure the longer side of the chip:
As you can see from the diagram above I could measure two different lengths. Therefore from my preliminary work I have decided to measure the loss or gain of weight to be more precise.
Fair Test
To keep a fair test I will keep all of my variables constant apart from the concentration of the solution. In my preliminary I changed my variables by not keeping the same length of the potato or the same volume of concentration which made my results inaccurate therefore in the investigation I plan to keep the volume and the potato chip exactly the same volume and length. I will keep the temperature constant throughout this experiment (something I did not measure during my preliminary) as an increase in temperature will increase the rate at which osmosis occurs, thus making my results inaccurate and finally to make sure my results are accurate and more precise I will repeat each experiment with the different concentrations at least 3 times. This will give me an average of my results thus making them more accurate. I will also leave the potato chips in the solution for the same amount of time as in my preliminary I did not do this and this could be a cause of any anomality so I will make sure that this does not happen.
Prediction
Gathering information from my background knowledge on osmosis and my preliminary work I have come to the prediction that as the concentration of water gets weaker that the potato chip will gain in weight. Therefore I expect that as the concentration of the solution gets stronger I will expect a weight loss in the chips, which means that I expect my graph to look like the one below:
Method
First of all we acquire 5 petri dishes for each of the solution and we then fill one petri dish with ml of 0.2 concentrated solution we do the same to the other petri dishes whilst remembering to in each petri dish we will add a different concentrated solution to it. We then cut one potato chip to a length of cm and weigh how heavy this is. Making sure we have recorded the initial weight of the potato chip we place one potato chip into each of the petri dishes filled with the different concentrated solutions. We next start the timer and leave them in there for mins. After the potato chips have been in the solution for mins we remove them at the same time wipe down any excess water and then re-weigh them. Do this 3 times with each solution (obviously not using you old solution again but replacing it with the same concentration but of a new solution). Once these results have been recorded in a table we will be able to find out the gain or loss in weight and therefore the percentage of weight gained or loss. With these results we can plot a graph of weight loss/gain against the concentration of each solution.
Apparatus
In this experiment we will be using the following apparatus:
4 potato chips
potato borer
China Tile
Distilled water (0.0 mol/dm3)
Various concentrations of sugar solution
Measuring cylinder
Weighing machine
Beakers
Petri-dishes
Safety
I’ll be wearing at all times safety googles uincase some of the liquid splashes into my eyes, I will also wear an apron to protect my clothing from any spillages. I will also take great care when using the borer, using it only on the white tile so that I don’t damged the ben and I can get a cleaner cut.
Observations
In my experiment I took used 6 different concentrations of sugar solution from a range of 0.0 mol/dm3 – 0.5mol/dm3 going up in 0.1mol/dm3. I put 4 potato chips in each concentration thus taking four reading to work out an avearage making my result more accurate. The potato chips before they went in the solution were weighed and then were weighed after they had been in the solution for 30 minutes. The scales gave reading of up to 2 decimal places. Before being weighed each potato chip was dried with toilet paper and with my results I came up with a table to show the percentage weight change:
Analysing
From my results table and graph it is evident that the potato chips have either gained or lost in weight, therefore this must mean that the concentration of a solution must have an affect on osmosis. Most of the pieces in the solutions of high concentration have lost weight and most of the potato chips in the low concentrations have gained weight. Therefore I can conclude that my experiment confirms my prediction that the greater the concentration of sugar the more weight and water the potato chip loses. My graph indicates this to me because as the concentration increases along the x axis the weight along the y axis goes down.
The line of best fit on the graph passes through x-axis when the concentration of salt solution is at about 0.09 moles/dm3. When the line passes through the x-axis at that point the rate of intake of water is equal to the rate at which it is released.Therefore the concentration of salt within the potato cells is approximately 0.09 moles.
In the solution of just water or a very weak solution where there is more water present on the out side of the cell than inside it, water has been taken in which has thus increased the mass of the potato. This is because the water has diffused through the permeable membrame of the cell. The water molecules have moved through the partially permeable membrame at a faster rate than they moved the other way. Therefore the potatoes in the solutions of sugar concentrations of 0 moles and 0.1 moles, an increase in weight has occurred. This is down to osmosis, the movement of water molecules from an area of high concentration to an area of low concentration.
In the solutions where there was a greater percentage of salt there was a decrease in the weight of the potato pieces. This means that more water left the cells via osmosis than went through the partially permeable membrane.This is due to the fact that there was a greater concentration of water inside the cell to that of outside the cell. Since osmosis is the movement of water molecules from an area of high concentration to an area of low concentration, water has moved out of the cell as the concentration of salt is greater on the outside of the cell. Here it is evident that Plasmolysis has occurred. As the water moved out of the cell the cell membrane pulled away from the cell wall and the cell has become flaccid. The potato chips in the solutions of 0.4 moles and 0.5 moles have lost more water and more weight than those in the weaker solution of 0.2 moles and 0.3 moles because the diffrence in water percentage between inside and outside the cell is greater when the potatoes are in 0.4 and 0.5 moles solutions.
I do not think that my graph would ever had taken the shape of a straight line. At the positive end, when the potatoes have gained weight, I would expect the line to eventually curve off vertically. This is because once the cell membrane starts to push against the cell wall, the tugor pressure will reach it’s maximum and the cells will not be able to take in any more water. At the other end of the line, I would expect the line to eventually curve off horizontally. Once the cell becomes flaccid and the cell membrane collapses, no more water would be able to escape.
I did predict that the stronger the solution the more the potato chips would lose weight. I believe my results have proven this.
Evaluation
I am pleased with the way my experiment has gone, I did not come across too many problems and most of my points on the graph were close to the line of best fit. Therefore I am happy with the accuracy of my results.
I think my method was adequate for producing accurate results I could still improve it. I could’ve also tried to measure the potato chips more accurately when cutting them.
I feel my investigation on the whole was a sucsess. I achieved reliable and accurate results. however I could increase my confidence in my conclusions by repeating the experiment with another carrot to back it up. But on the whole my experiment matched my prediction and I am happy about my results and the way I conducted it.